Cell membranes provide a highly effective barrier against toxic substances and environmental stresses. The core structural element of biological membranes is a lipid bilayer. Embedded within this bilayer are proteins, many of which transport ions and water-soluble molecules across the membrane.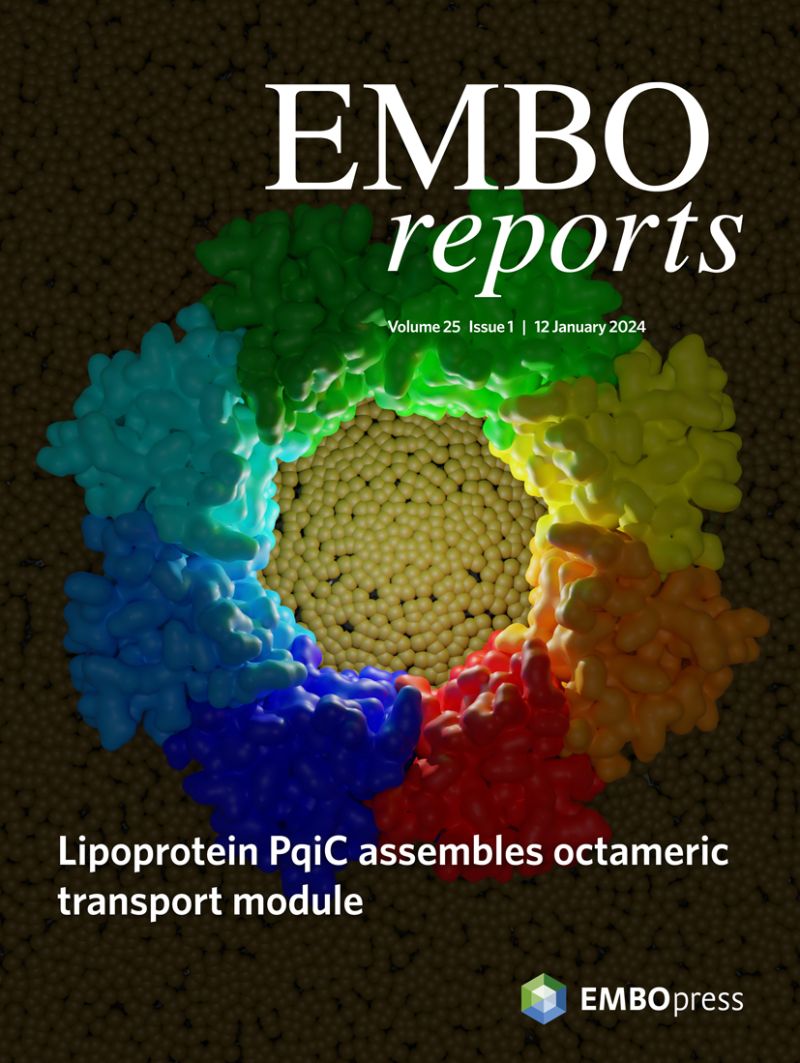
Unlike other organisms, Gram-negative bacteria have two membranes surrounding the cell, this is one of the main reasons why they are highly resistant to antimicrobials. The transport pathways required to form these membranes have been extensively studied, but surprisingly it still remains ambiguous how lipids are transported between the membranes. MCE proteins (comprising one or more mammalian cell entry domains) have been implicated in the transport of lipids in both mycobacteria, which causes tuberculosis, and E. coli, a common bacteria known primarily for causing gut infections. E. coli contains three MCE domain-containing proteins, MlaD, LetB and PqiB, which have all become of principal interest as components of transport systems.
In E. coli PqiB, forms part of the PqiABC system, a pathway comprised of three components: PqiA, an integral inner membrane protein, PqiB, which spans between the two membranes and an outer membrane lipoprotein, PqiC. Interactions between these components remain uncharacterised. However, once assembled, this trio of Pqi proteins is speculated to provide a transport system between the two membranes of the envelope. In this study, the team focused on PqiC using X-ray crystallography to solve its structure, revealing its biological assembly to form an octameric ring.
Using OFFSPEC, the team then used neutron reflectometry to investigate the nature of the membrane interaction between PqiB and PqiC. They found that the PqiC octomeric ring embeds partially into the inner section of the Gram-negative outer membrane. These findings support a model in which PqiC acts as an anchor at the outer membrane for the C-terminal element of PqiB, limiting its flexibility, whilst potentially allowing its movement within PqiC.
“This research provides another piece of the membrane biogenesis puzzle in gram-negative bacteria, providing key insight into the structure of a protein complex implicated in lipid transport," explains corresponding author Dr Tim Knowles, Reader in Structural Biology and Director of the Midlands Integrative Biosciences Training Partnership at the University of Birmingham. "Central to this research is the ISIS Neutron & Muon Source, its incredible capabilities provide unique tools for studying the complexities of membrane biogenesis in ways never conceived before, we will continue to work closely with them to push what is possible with neutron science."
This paper is available to read below and also featured on the issue cover of EMBO Reports for January 2024.
An octameric PqiC toroid stabilises the outer-membrane interaction of the PqiABC transport system | EMBO reports (embopress.org)