Oil and gas from shale and tight sand reserves account for over half of U.S. production. Following the initial oil/gas retrieval, carbon dioxide (CO2) injection could be used for enhanced oil recovery. As well as this, the shale formations could also be potential stores for CO2 as part of carbon capture and storage efforts. It is important to understand how much CO2 could be stored in the shale, and how it moves inside the rock structure, before relying on these techniques for economic benefit or CO2 mitigation targets.
Thanks to the instrument's ability to provide information on both the structure of fluids in porous materials, and the structure of the mesoporous material itself, NIMROD is well suited to investigating the behaviour of CO2 in these structures.
A previous experiment, featured in our 2018 Annual Review, found that large numbers of the micropores in shale are closed, and therefore unavailable for CO2 storage. Building on this work, a group from the U.S. Geological Survey (USGS) brought two samples to NIMROD that were mineralogically different, to see if this variation had an influence on the rock's interaction with CO2.
One sample studied was composed of relatively high amounts of quartz, moderately high amounts of clay and low amounts of carbonate, whereas the other was composed of relatively equal amounts of quartz, carbonate and clay. Both samples were loaded with high pressure CO2 at room temperature; the conditions that would be used in the field, with one sample tested over a range of pressures from 20 to 50 bar.
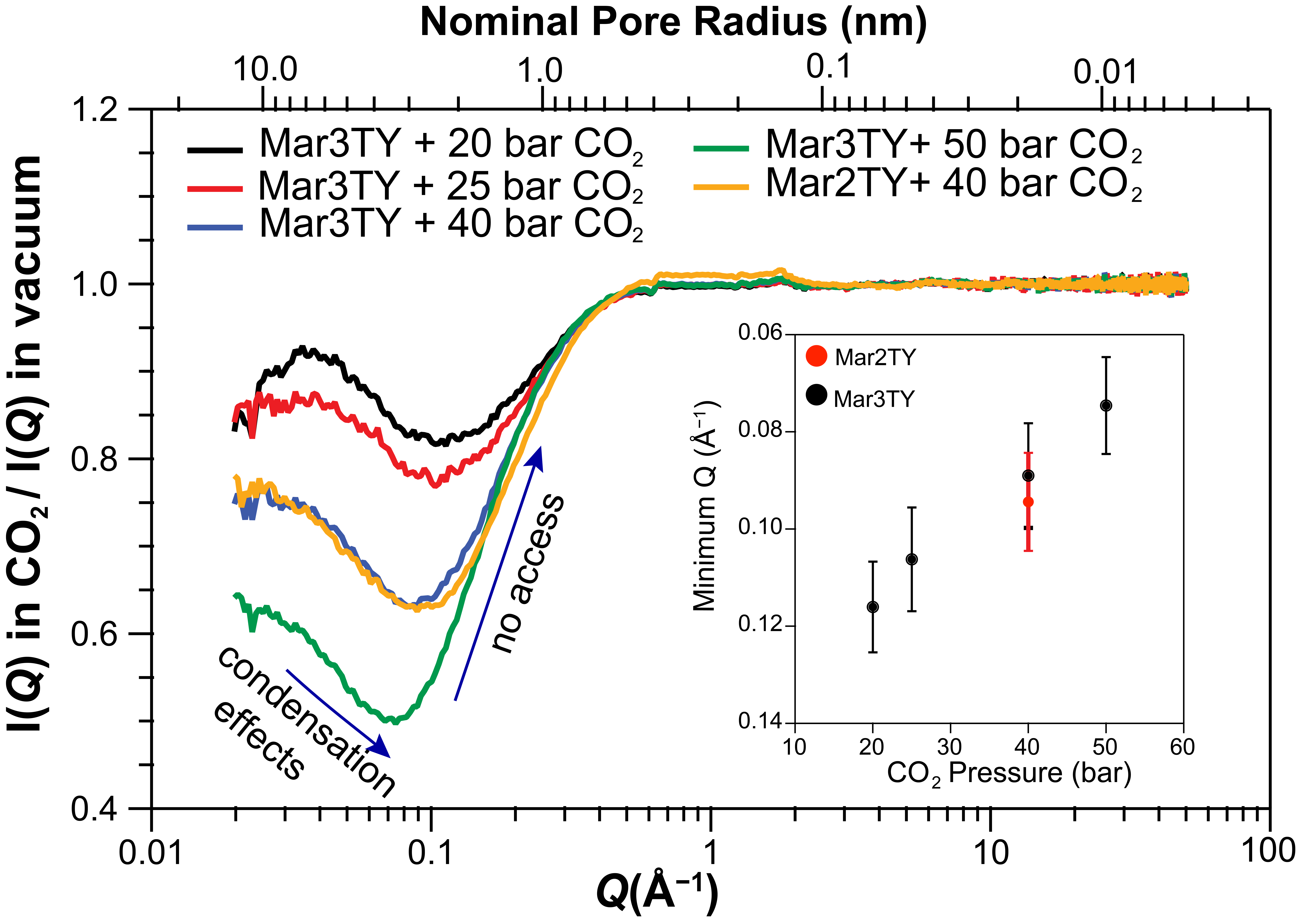
Ratio of NIMROD scattering intensities for Mar2TY and Mar3TY samples in the presence of pressurized CO2. Inset: Scattering ratio minimum vs CO2 pressure for the Mar2TY (red circle) and Mar3TY (black circles) samples investigated in this study. Error bars correspond to the propagated error at the 2s level of uncertainty. Reprinted with permission from Energy Fuels, 2020. Copyright 2020 American Chemical Society.
There was only a small difference between the two samples, indicating that, for these samples, the CO2 storage occurs in the part of the shale made of organic material, known as kerogen, rather than the inorganic clay, quartz or carbonate.
At the lower pressures studied, some of the gaseous CO2 condenses in pores as large as 6 nm, and there is CO2 condensation within some of the larger pores at higher pressures. As the pressure increases up to 50 bar, CO2 is also seen as a liquid in the smaller pores (0.5-3.5 nm). CO2 is not seen in any pores smaller than this, which the researchers think is due to the molecule itself being too large to enter the 'throats' of the pores, even if the pores themselves are big enough to contain them.
This formation of liquid CO2 in the smaller pores, and in the pore throats, would prevent the gaseous CO2 from filling the porous structure, as would be needed for both enhanced oil recovery (EOR) and CO2 storage. This may explain the low levels of EOR seen in field simulation tests when compared to model systems.
Further Information
The full paper can be found online at: DOI: 10.1021/acs.energyfuels.9b03744
View other science highlights from NIMROD.